Antibody-dependent cellular cytotoxicity targeting CD4-inducible epitopes predicts mortality in HIV-infected infants
BACKGROUND:
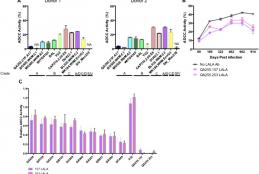
Antibody-dependent cellular cytotoxicity (ADCC) has been associated with improved infant outcome in mother-to-child transmission (MTCT) of HIV-1. Epitopes of these ADCC-mediating antibodies remain unidentified. CD4-inducible (CD4i) epitopes on gp120 are common ADCC targets in natural infection and vaccination. We tested whether CD4i epitope-specific ADCC mediated by maternal antibodies or passively-acquired antibodies in infants is associated with reduced MTCT and improved infant survival.